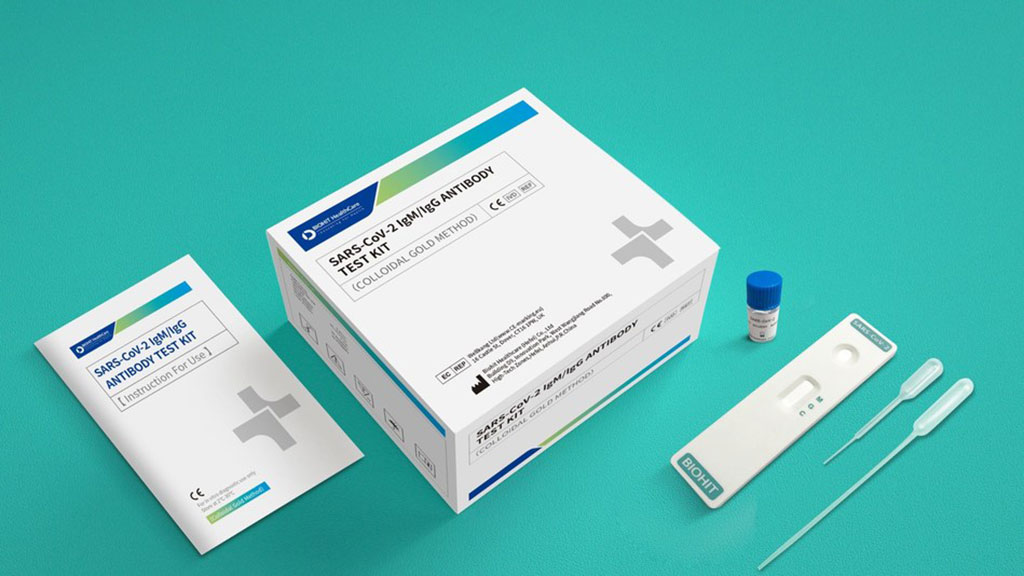
Covid 19 Antibody Test Kit Abbott - Covid-19 Realtime Info
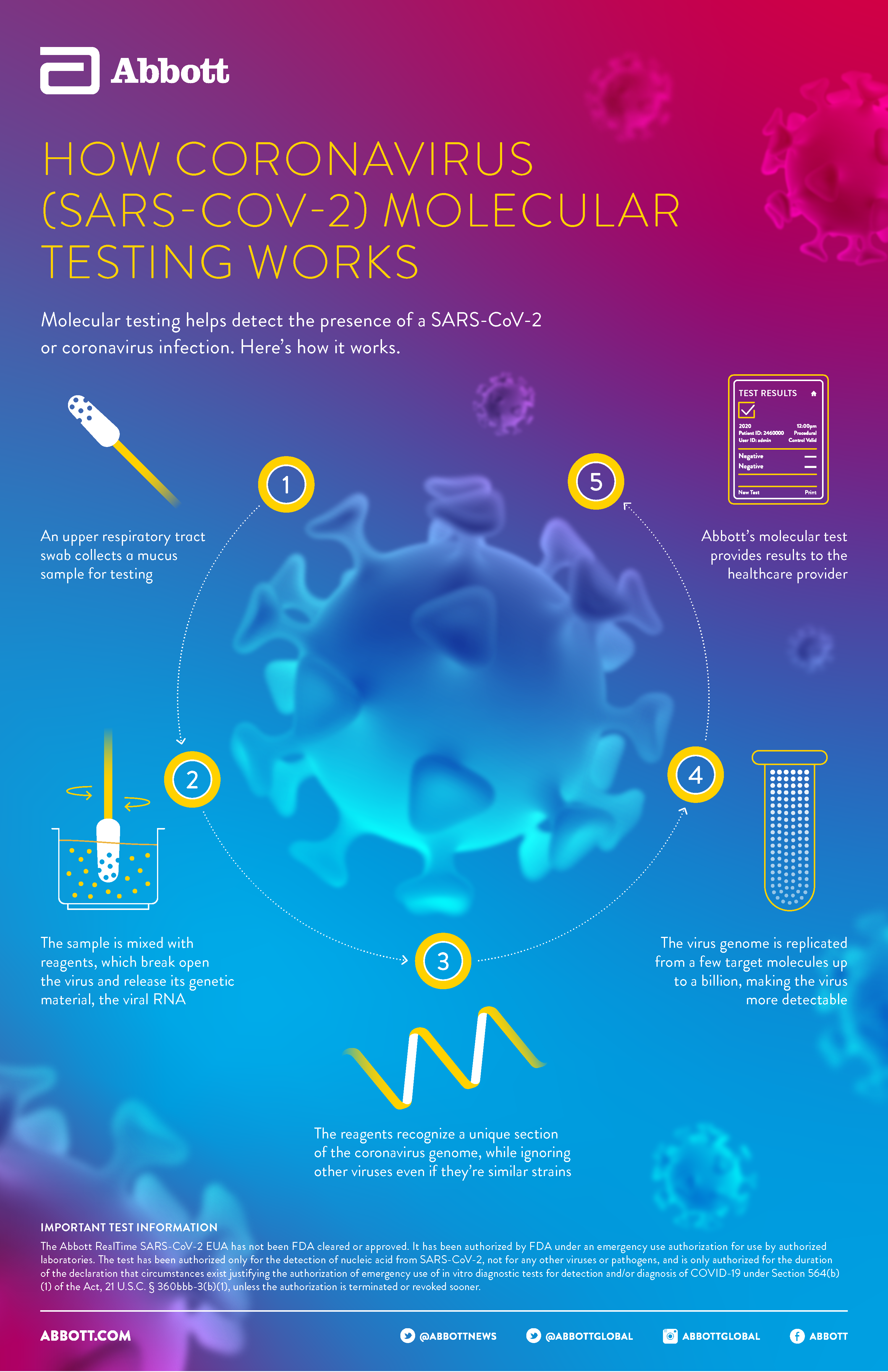
Abbott has also received euas for molecular tests that can detect current infections with covid 19.
:strip_exif(true):strip_icc(true):no_upscale(true):quality(65)/arc-anglerfish-arc2-prod-gmg.s3.amazonaws.com/public/I2VUSUXRGFDNRA3T6P2TPIA7Q4.jpg)
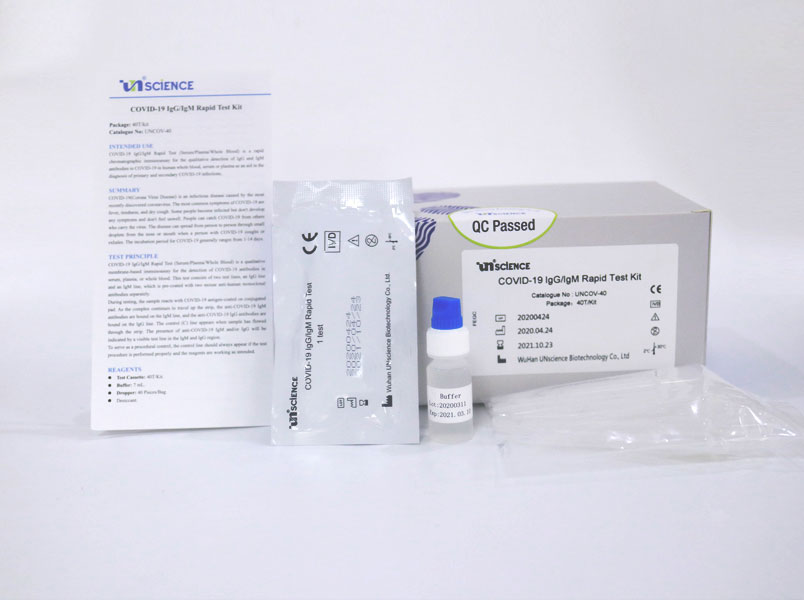
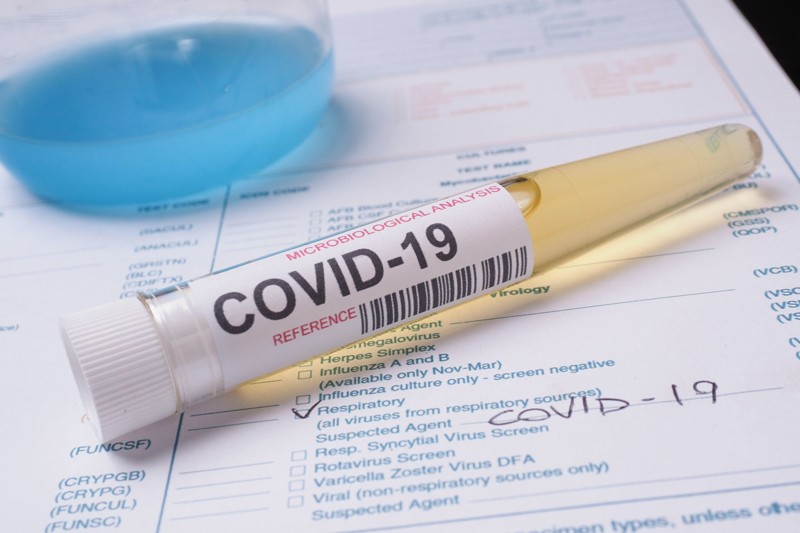
Covid 19 antibody test kit abbott. Abbotts architect sars cov 2 test. The abbott test made in europe can be used in machines already in labs. An automated test that can be used in labs and a test that can deliver. We are a us based early stage in vitro diagnostics company on the front end of the fight to combat the ravaging effects of the coronavirus.
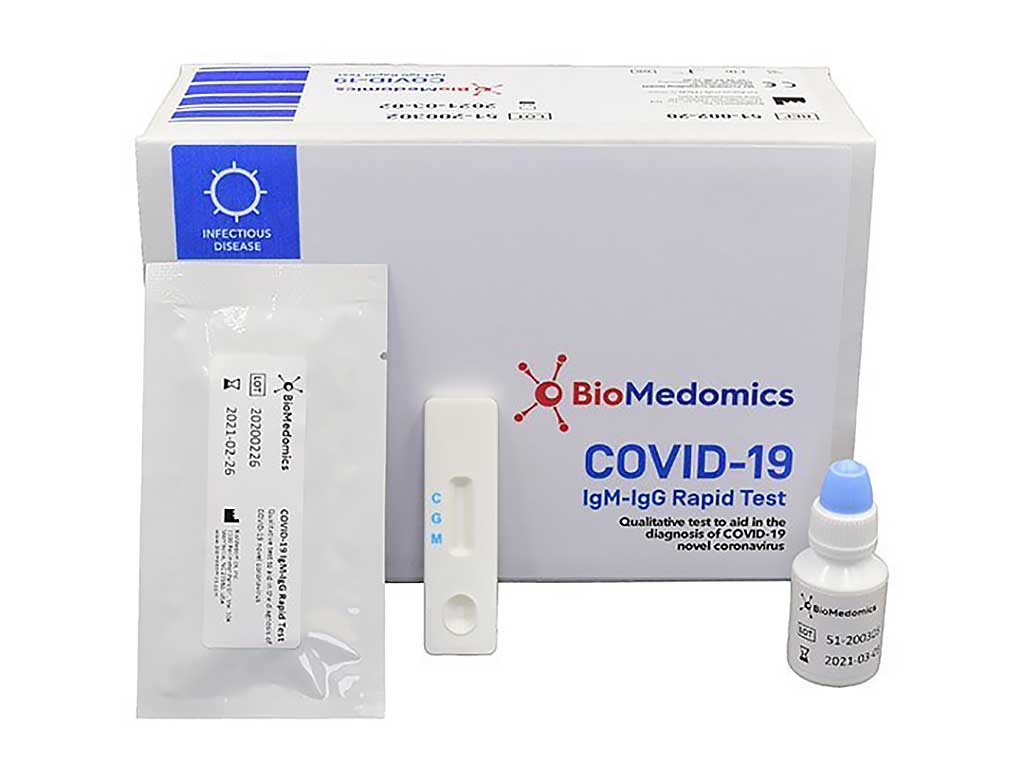
But with the covid 19 antibody test there are limited chances. Abbott immunoassay covid 19 igg immunity antibody blood test 49. Here are the details. Abbott who manufactured the test said it was never intended to be a home sampling kit.
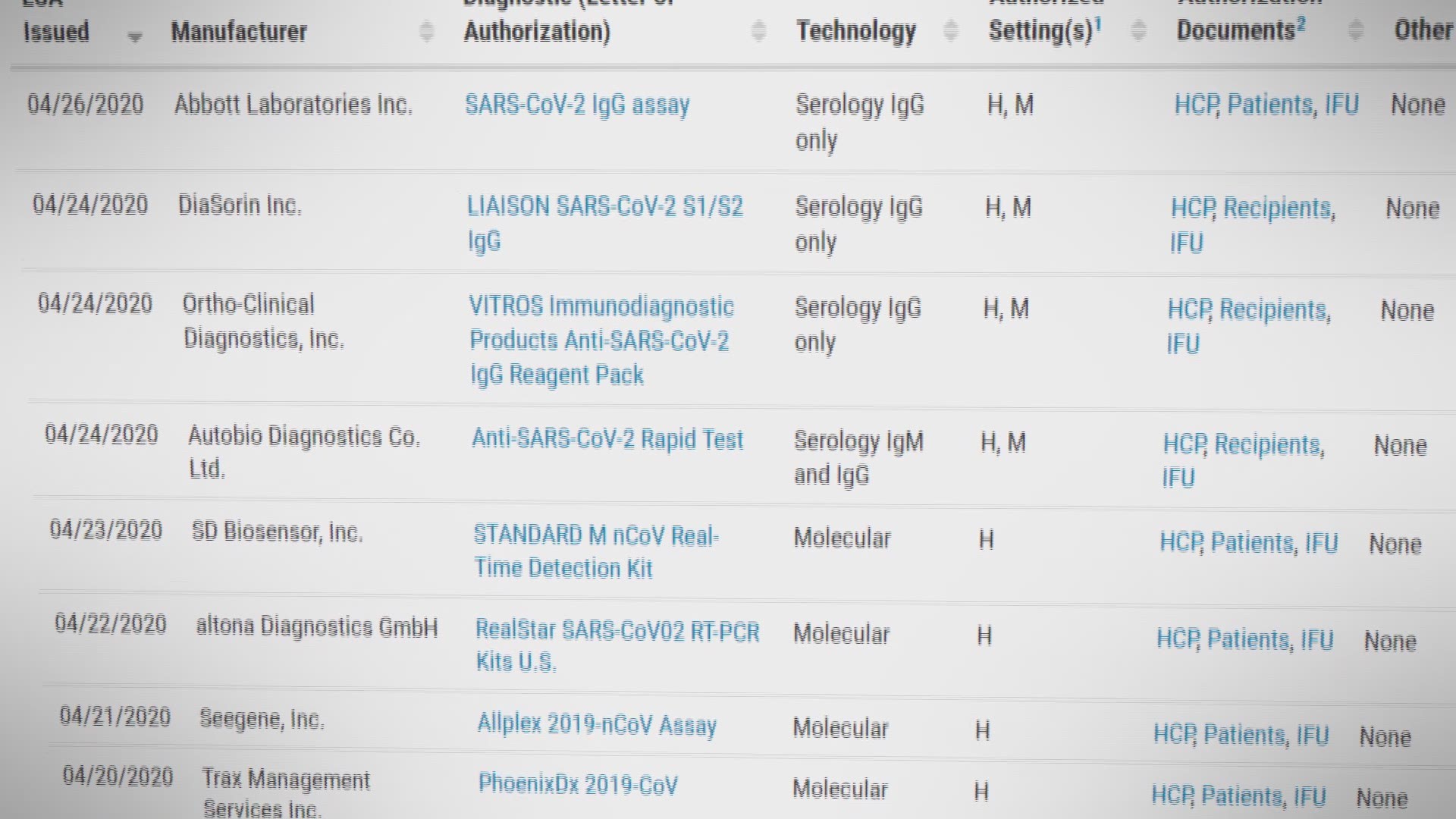
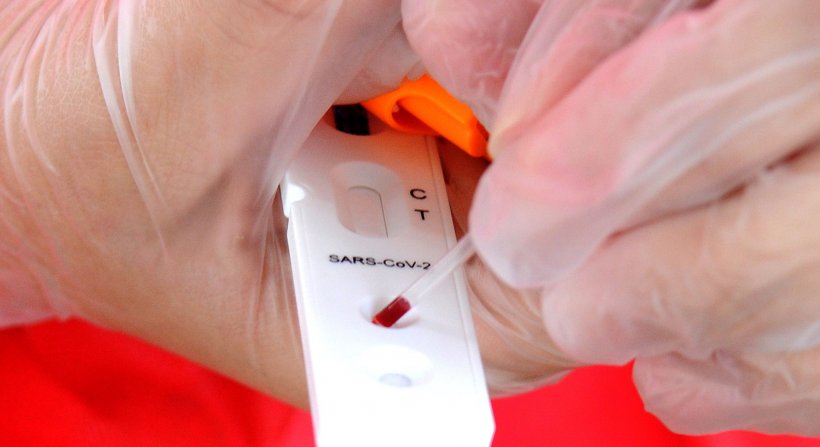
For two coronavirus testing kits. Two tests for covid 19 antibodies developed by the drug companies roche and abbott are highly specific but one was found to have lower sensitivity than was previously reported by the company evaluation by public health england has shown. Abbott has received emergency use authorization eua from the us. What makes this test so different is where it can be used.
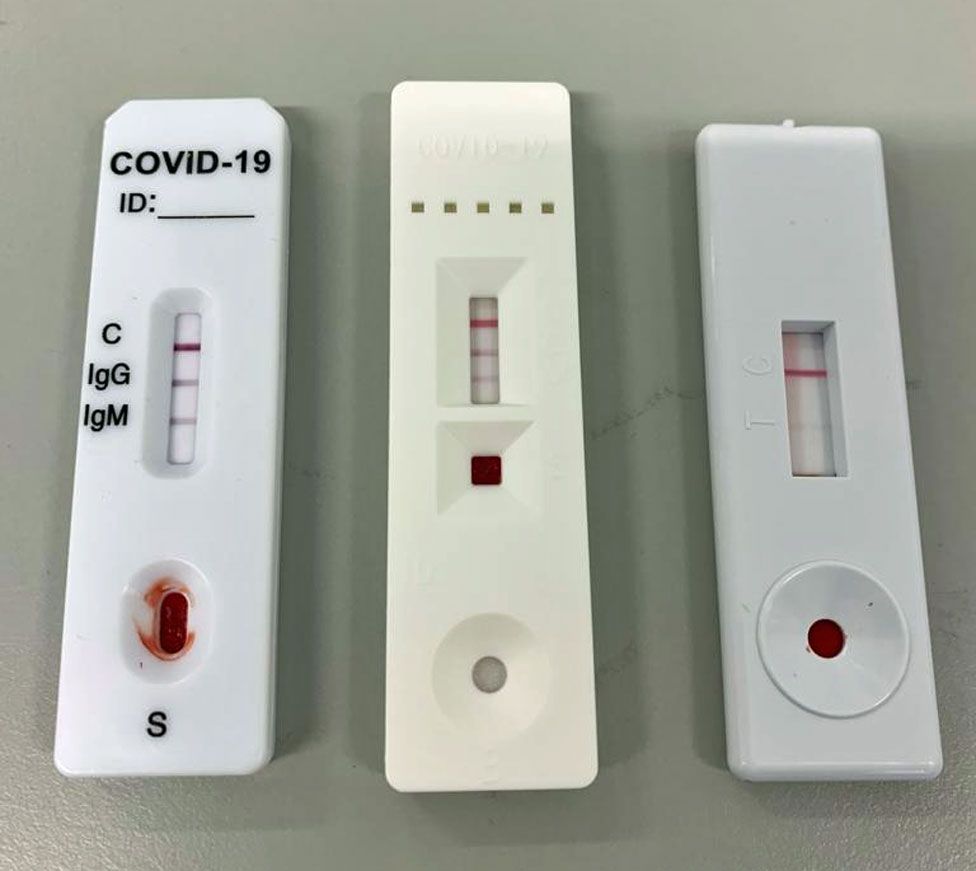
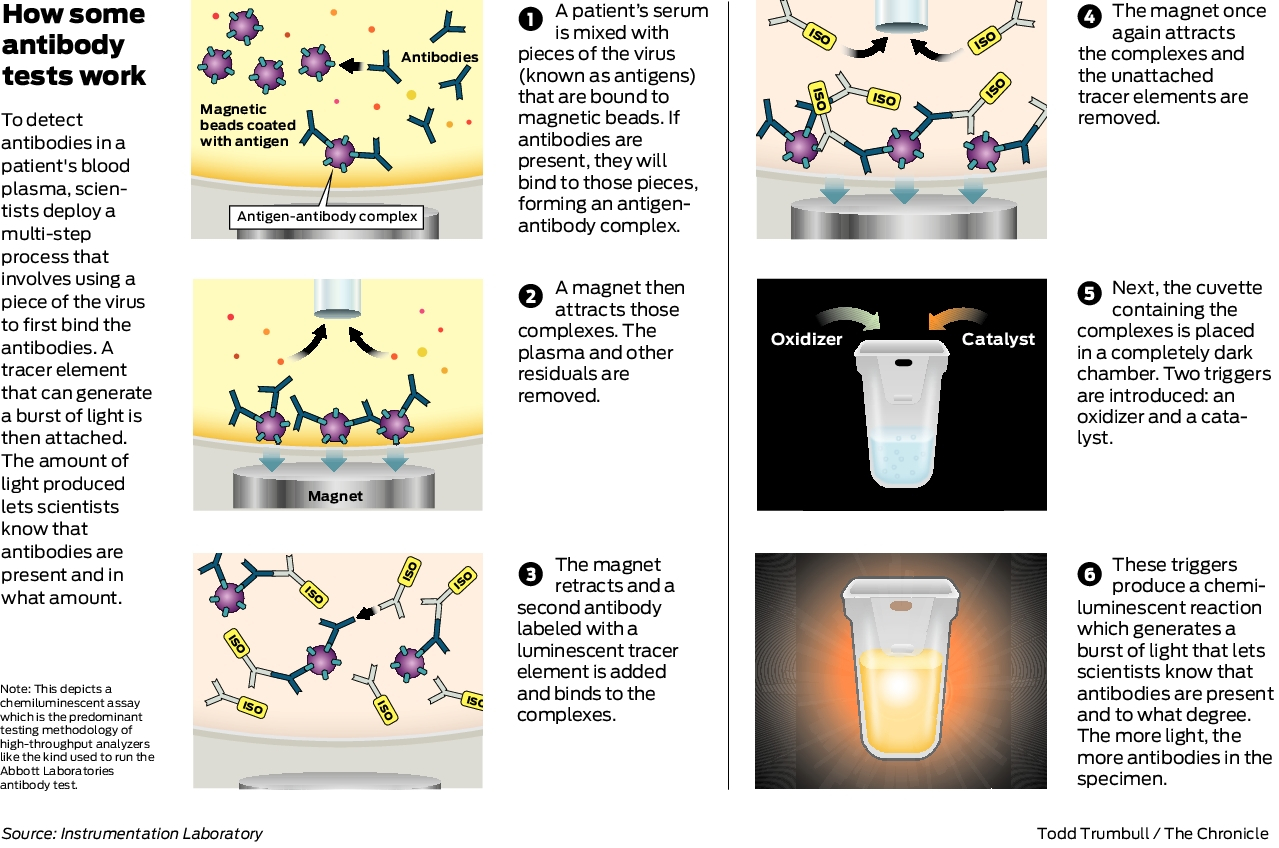
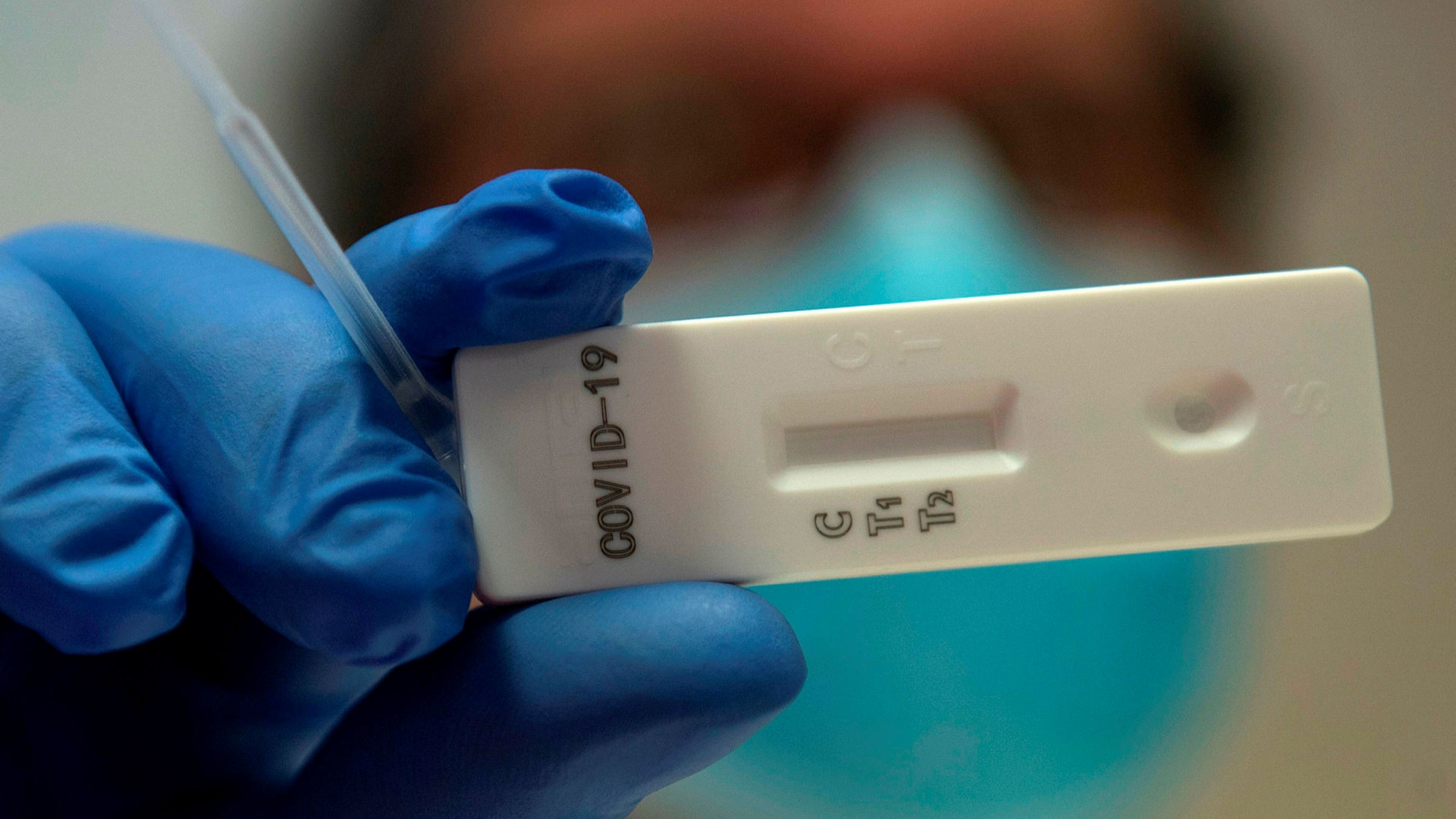
This new test is a serology test also called an antibody test and helps to detect the igg antibody to sars cov 2. Abbott launched its third covid 19 test and will start shipping in the us. It was also more than 99 per cent specific in identifying that 1070 negative samples did not contain covid 19 antibodies. Abbott had reported that its assays had a sensitivity of 100 the true positive rate 14 days after symptom onset.
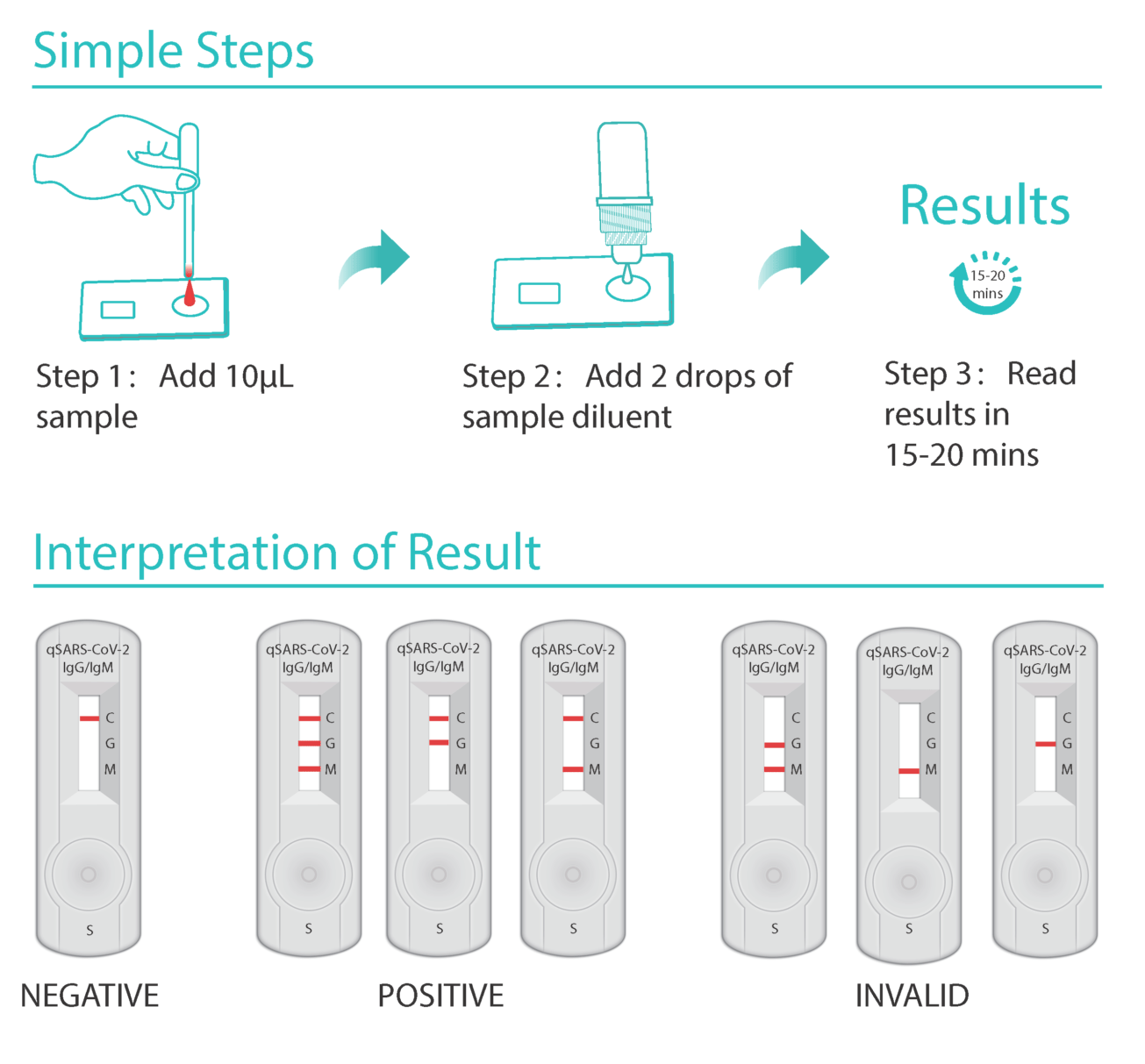
Book on line as gp blood test. Detecting these igg antibodies will help determine if a person was previously infected with the virus that causes covid 19. Five million coronavirus antibody kits are on standby for nhs use after a second test was approved by health officials. This is the newly approved covid 19 venous blood antibody test that we as doctors have been waiting for to provide good quality evidence of immunity.
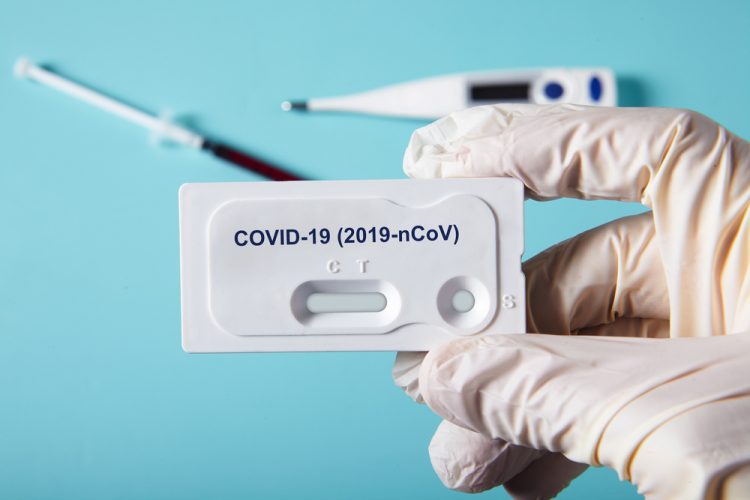
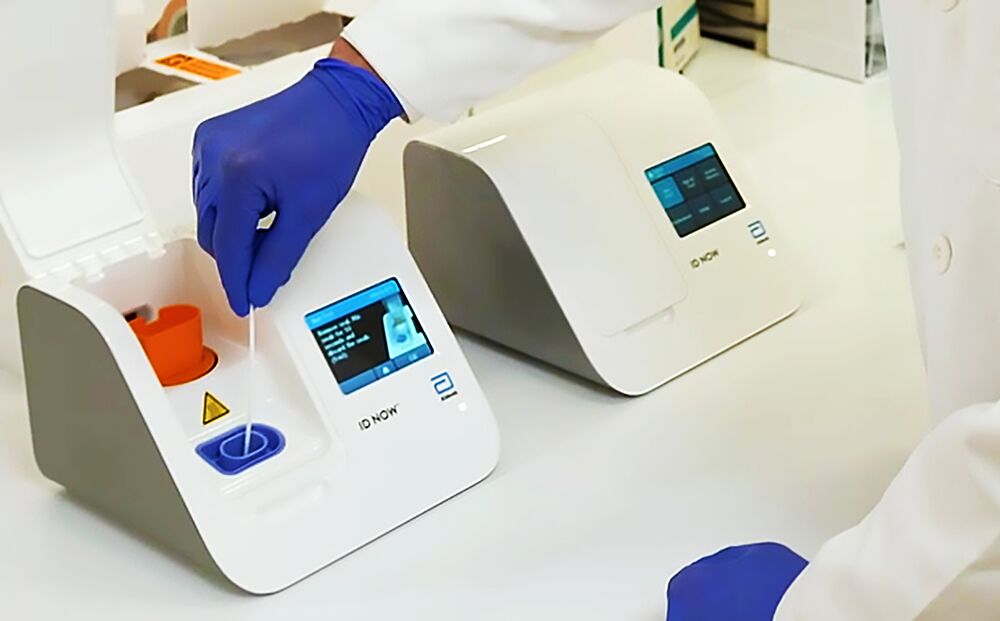
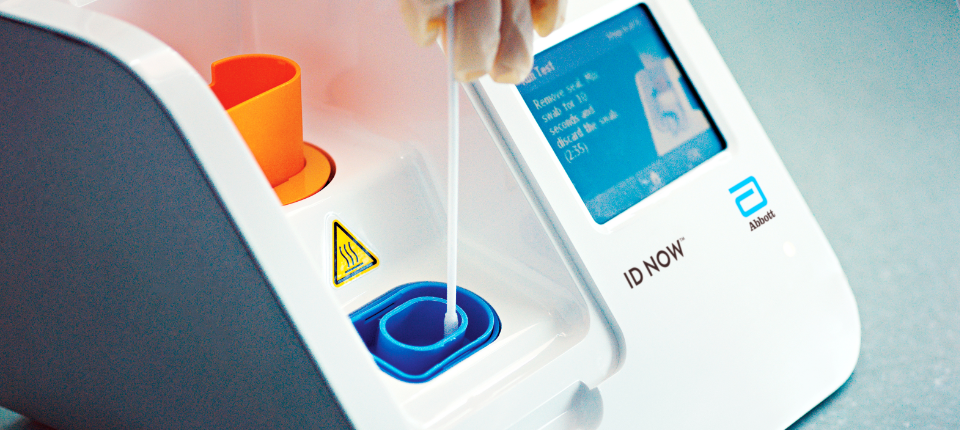
The new test produced by medical giant abbott has been given the. Food and drug administration fda for the fastest available molecular point of care test for the detection of novel coronavirus covid 19 delivering positive results in as little as five minutes and negative results in 13 minutes. Outside the four walls of a.








:strip_exif(true):strip_icc(true):no_upscale(true):quality(65)/arc-anglerfish-arc2-prod-gmg.s3.amazonaws.com/public/I2VUSUXRGFDNRA3T6P2TPIA7Q4.jpg)














/https://static.texastribune.org/media/files/0f33211f7bec164805c97e8fdac34eb2/Laredo%20Test%20Kits%20Lede%20TT%2002.jpg)






































/cdn.vox-cdn.com/uploads/chorus_asset/file/19856029/IDNOW_INACTION3_macro_300dpi_1200x628.jpg)