Covid 19 Antigen Test Sensitivity And Specificity - Covid-19 Realtime Info
A negative antigen test means that sars cov 2 viral proteins were not detected.
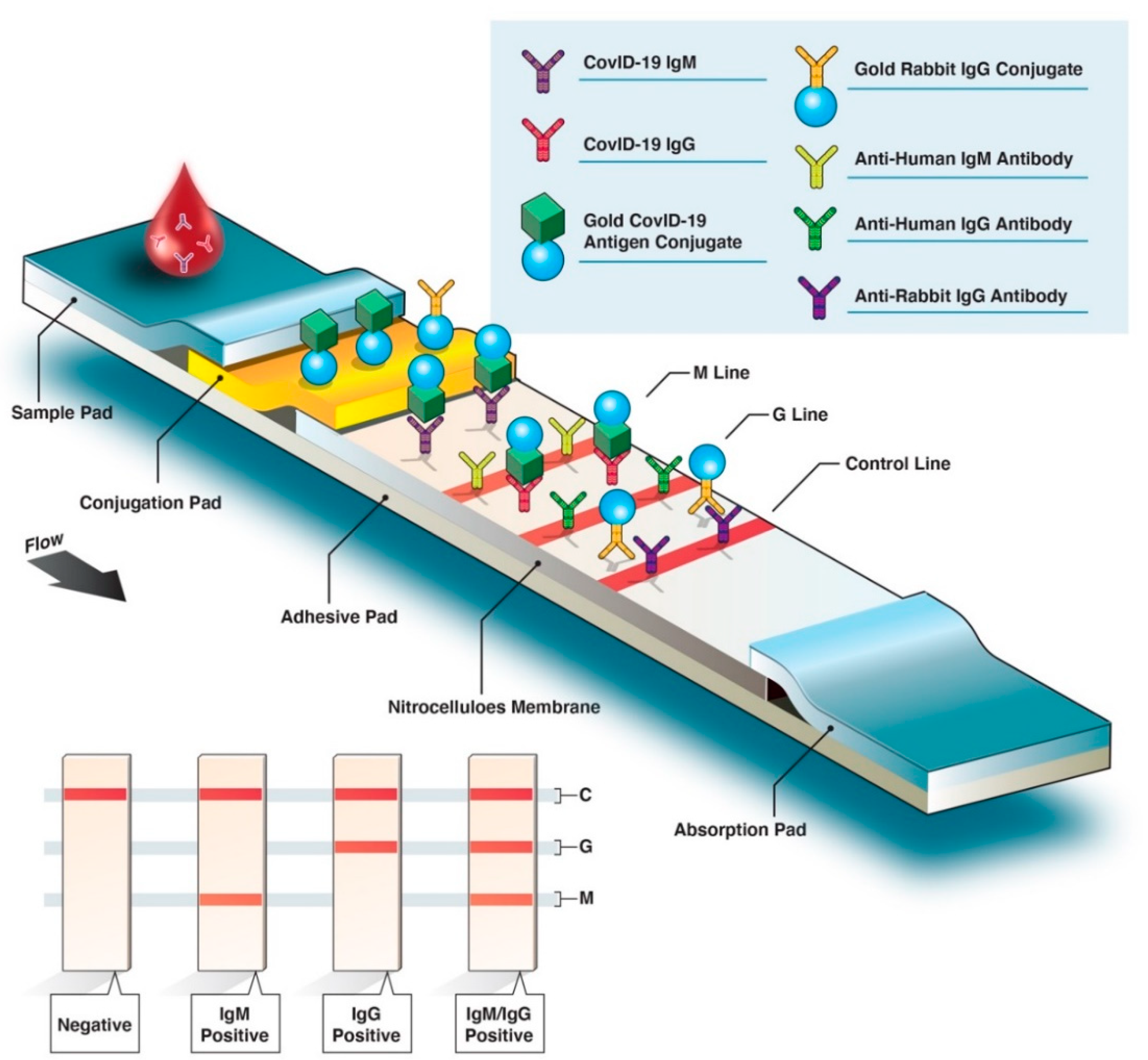
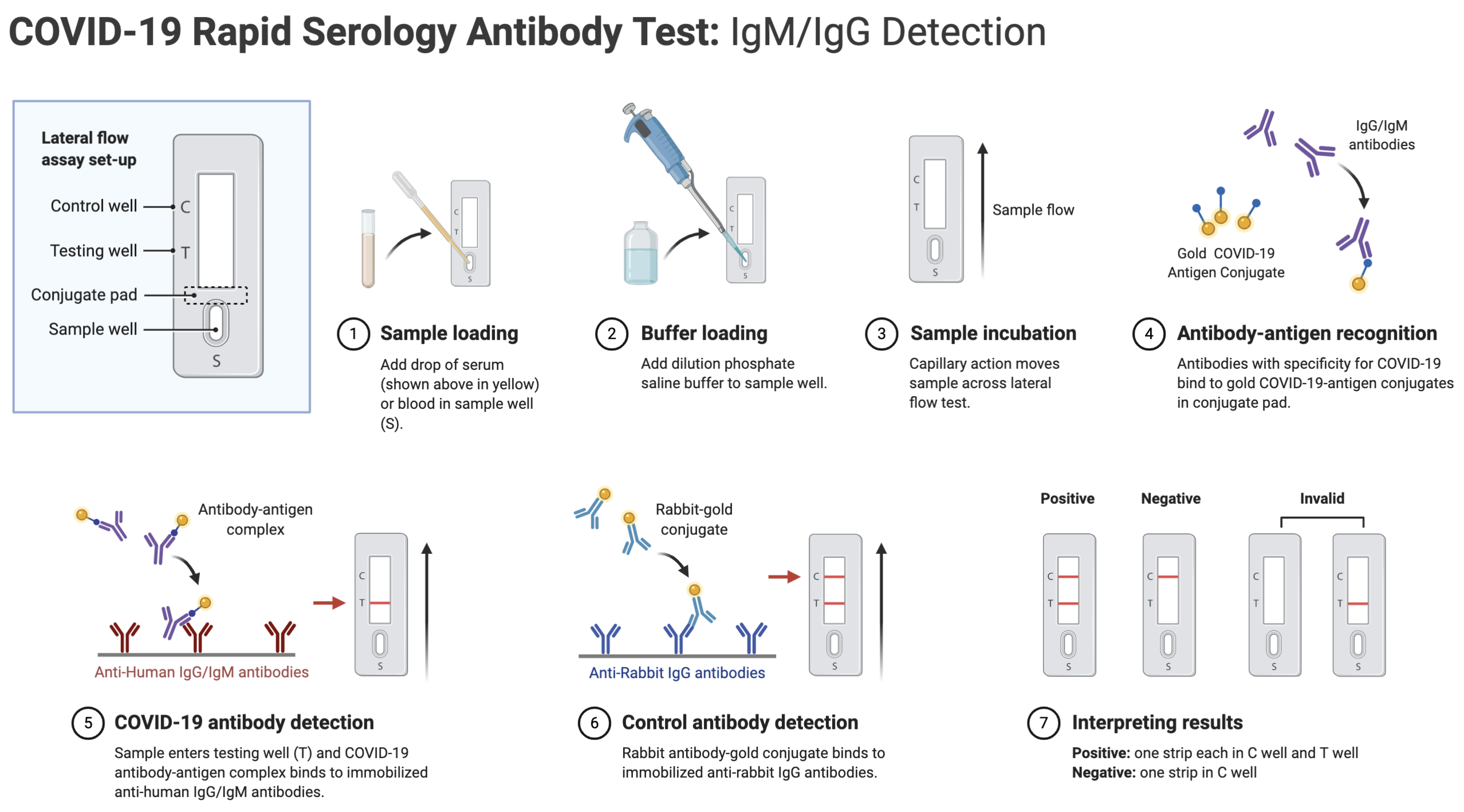
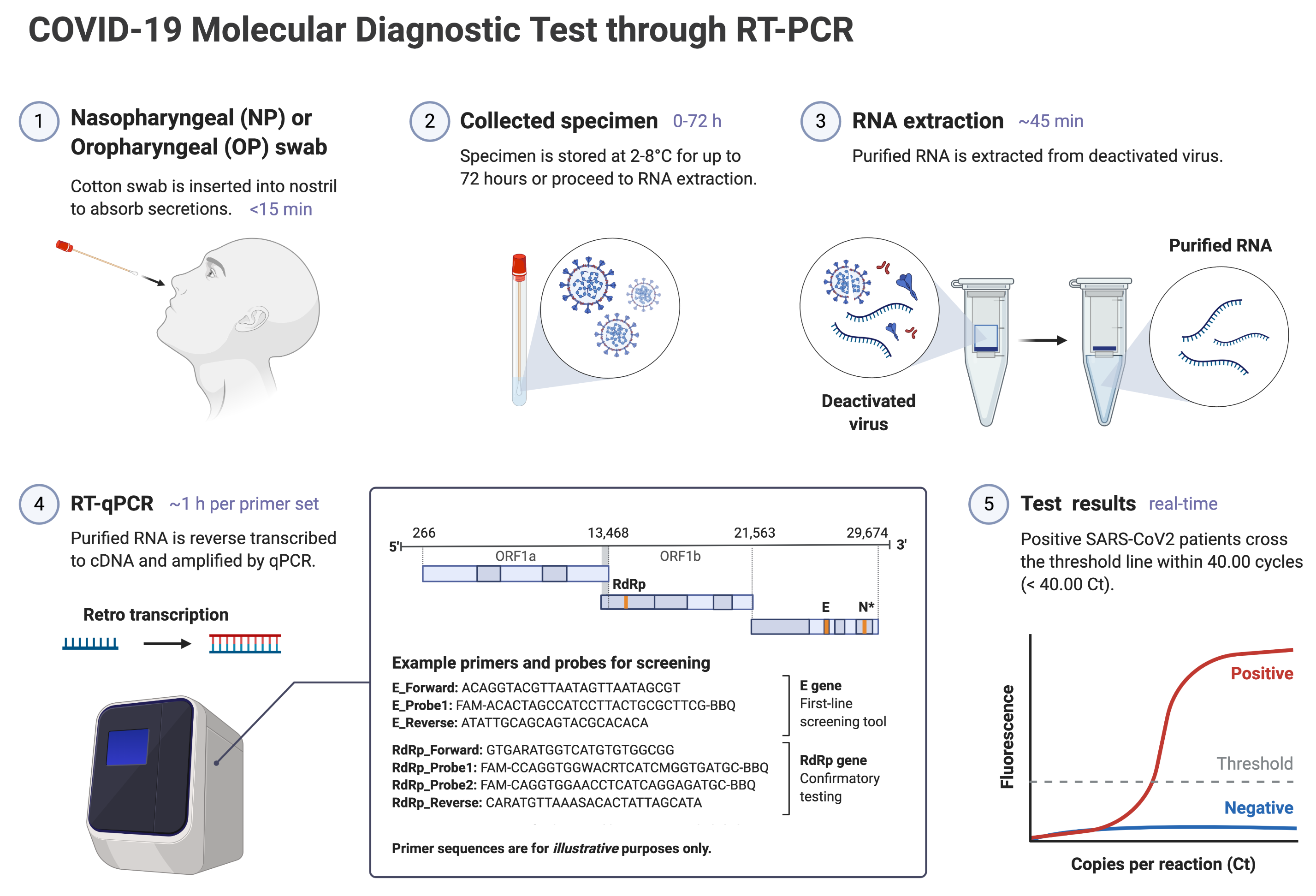
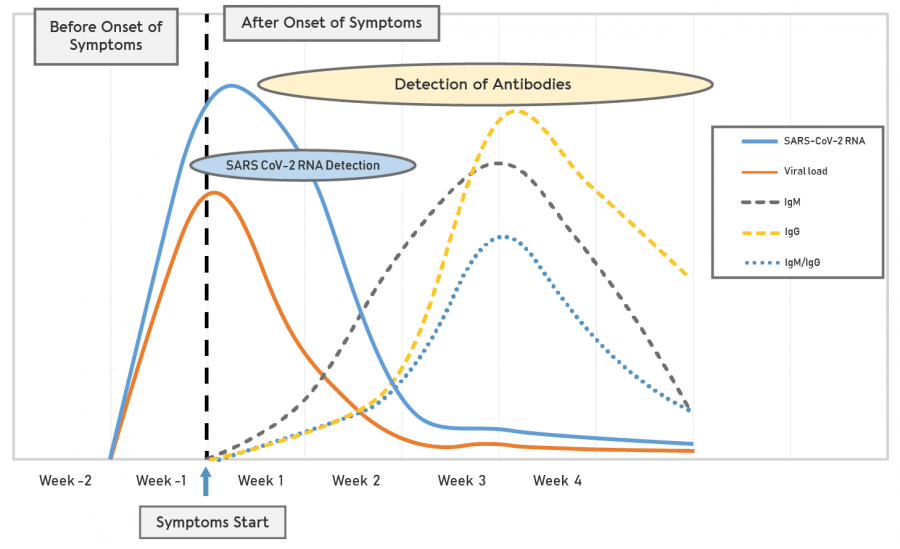
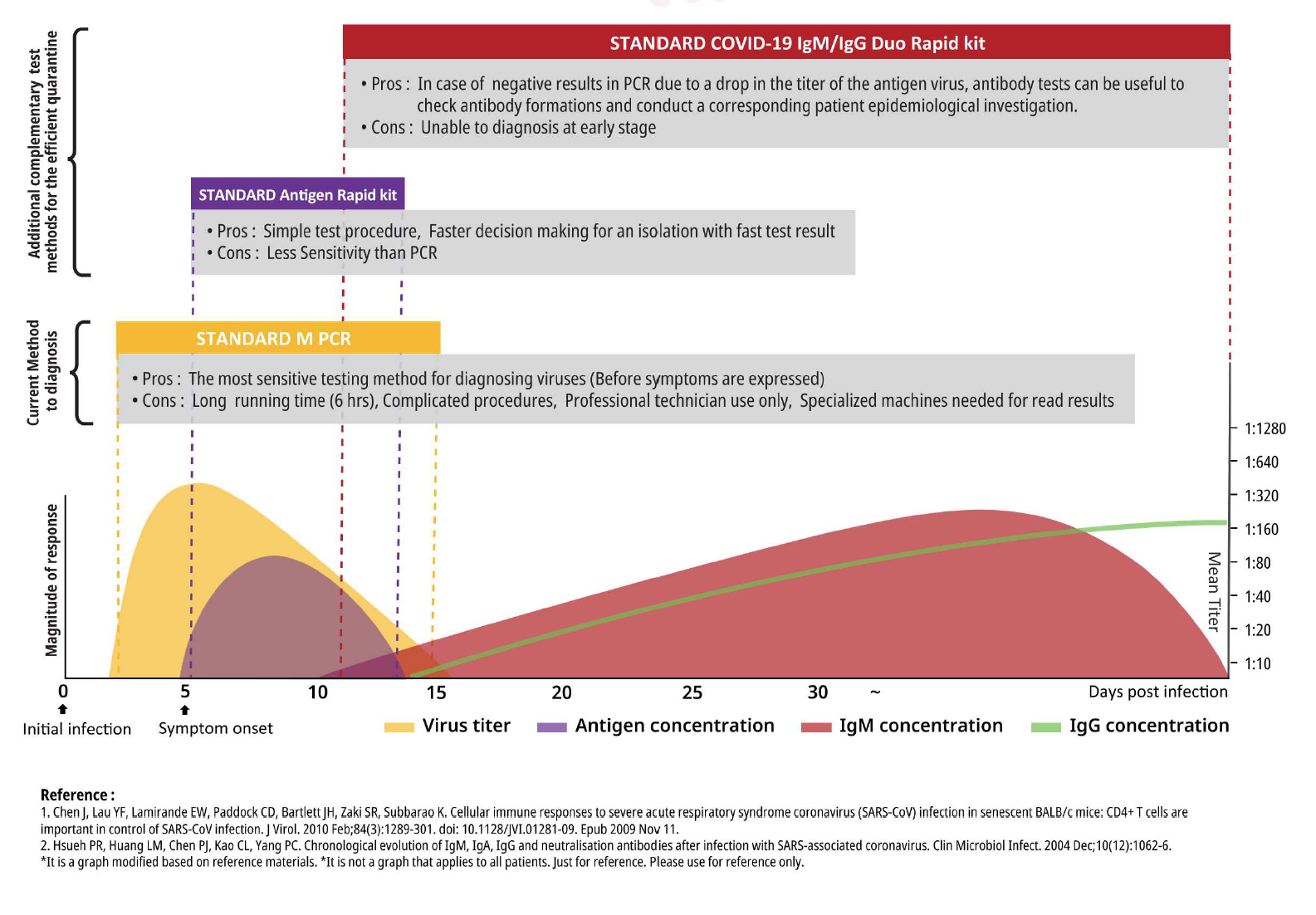
Covid 19 antigen test sensitivity and specificity. One type of immunoassay an antigen based test. Abbott had reported that its assays had a sensitivity of 100 the true positive rate 14 days after symptom onset. Researchers or clinicians collect samples from easy to reach areas like the nasal passage where the virus tends to replicate. It can be used to identify past sars cov 2 infection in people who were infected at least 1 to 3 weeks previously.
Note on sensitivity and specificity data. Molecular based tests for covid 19. However a negative test does not rule out covid 19. The covid 19 treatment guidelines panel the panel recommends that a molecular or antigen test for severe acute respiratory syndrome coronavirus 2 sars cov 2 should be used to diagnose acute sars cov 2 infection aiii.
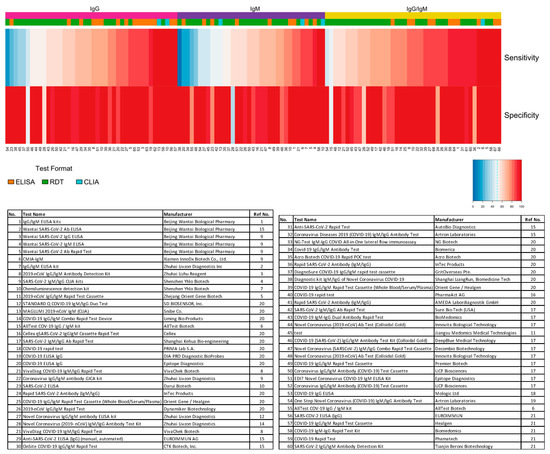
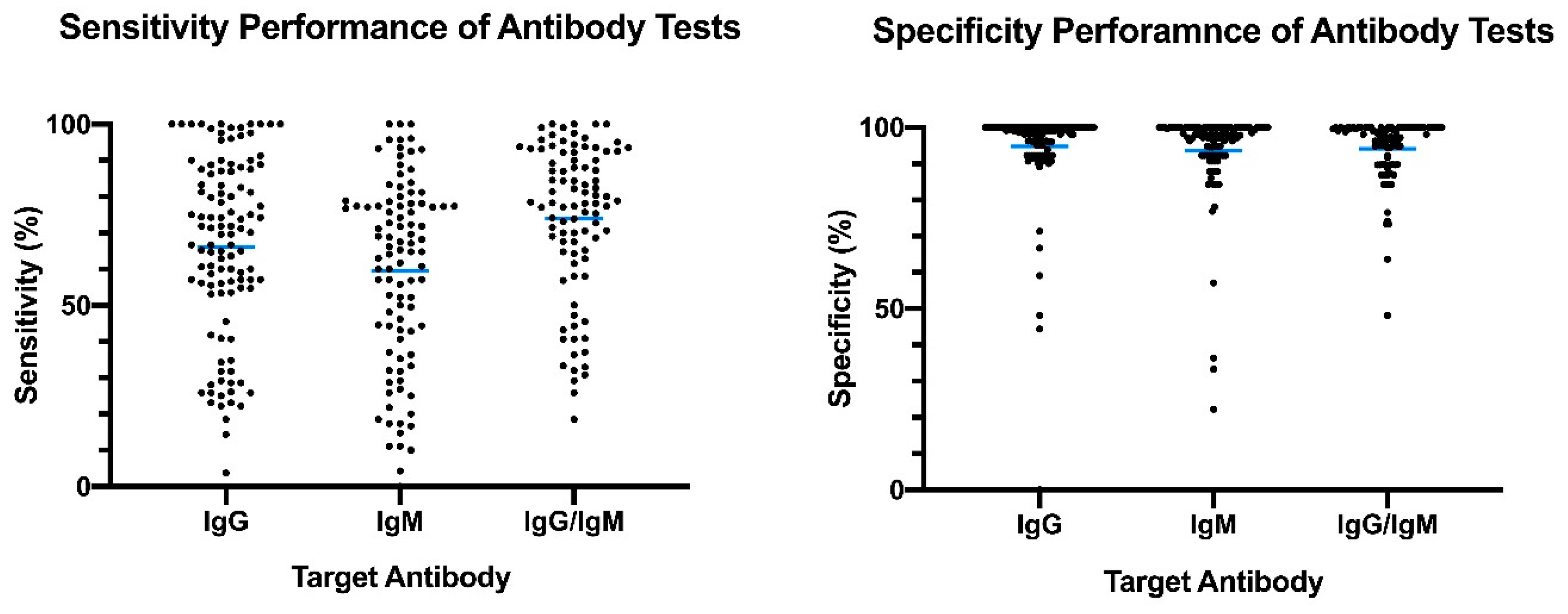
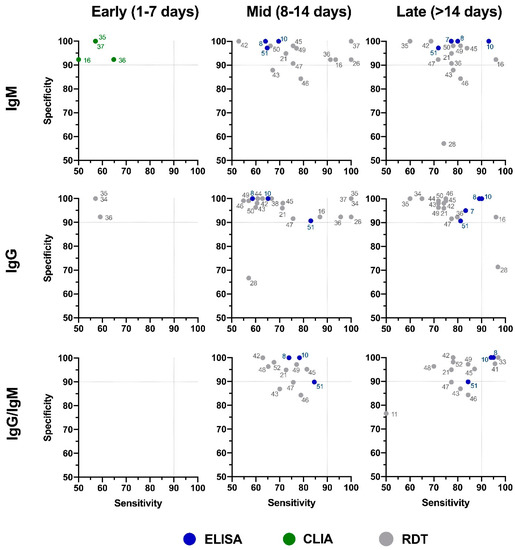
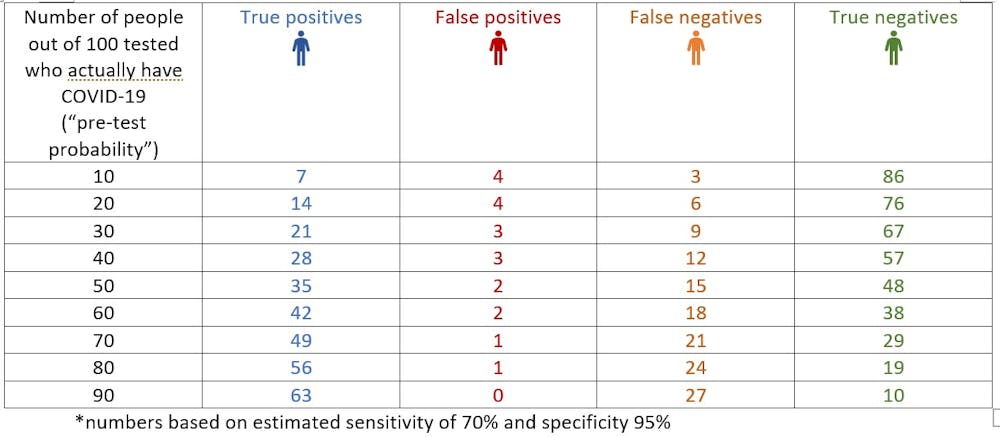
The panel recommends against the use of serologic testing as the sole basis for diagnosis of acute sars cov 2 infection aiii. A positive rt pcr test for covid 19 test has more weight than a negative test because of the tests high specificity but moderate sensitivity. Two tests for covid 19 antibodies developed by the drug companies roche and abbott are highly specific but one was found to have lower sensitivity than was previously reported by the company evaluation by public health england has shown. The specificity of the antigen test is reportedly around 99.
According to the who the sensitivity of similar antigen tests for respiratory diseases like the flu ranges between 34 and 80. That means a positive test result can be highly relied on to diagnose an infection. Covid 19 at the time of testing. Detects easy to find surface markers on the outside of the virus and avoids extraction and amplification steps.
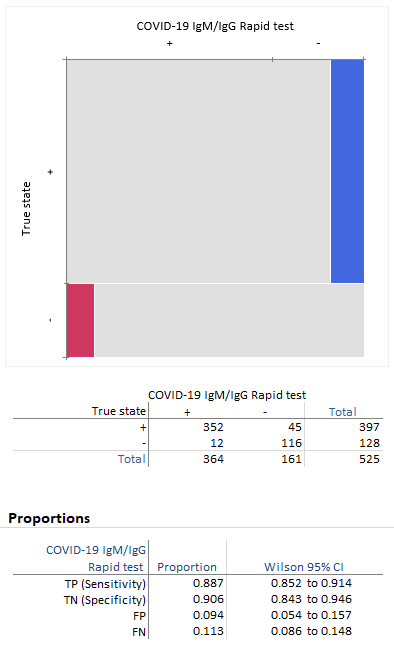
Sensitivity on the other hand is reported to be. Authorities in spain and britain ordered millions of covid 19 test kits. Clinicians should share information with patients about the accuracy of covid 19 tests. Cdcs serologic test has a specificity of greater than 99 and a sensitivity of 96 based on initial performance evaluations.
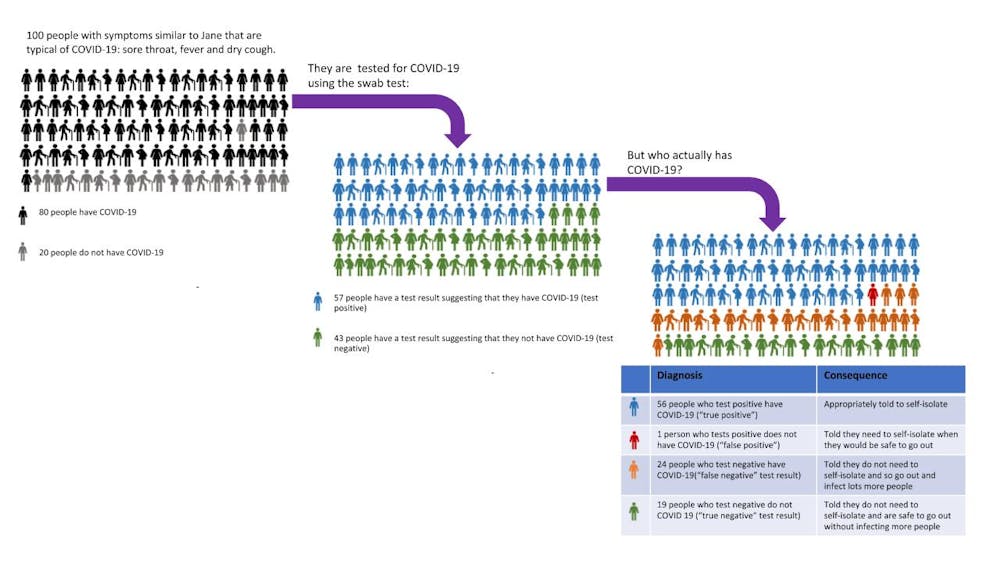
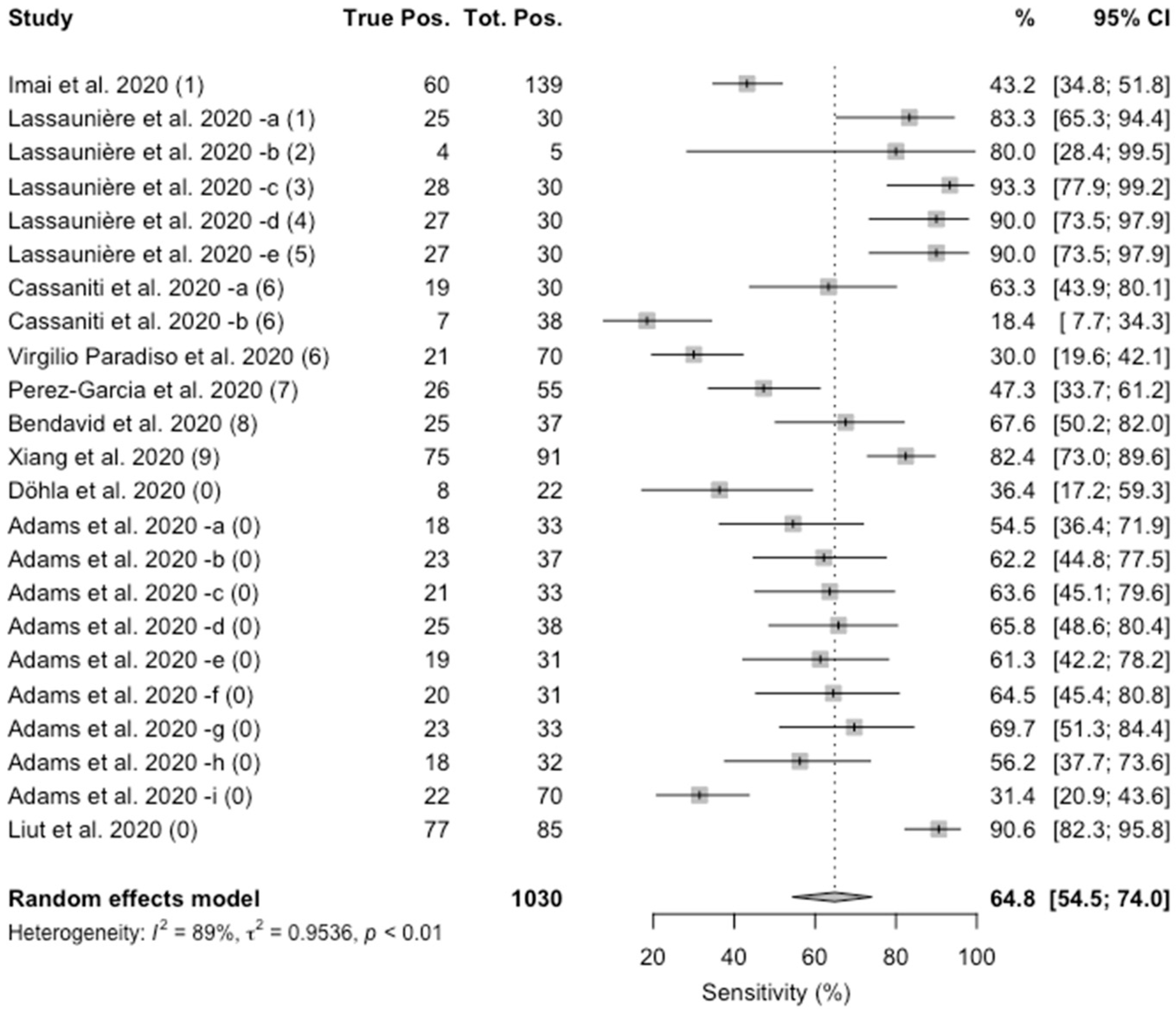
Sensitivity specificity instructions for use of the fda authorized assay the prevalence of covid 19 in that particular community positivity rate over the previous 710 days or cases per population and the. Evaluating the results of a rapid antigen test for sars cov 2 should take into account the performance characteristics eg. If there is still concern that a person has covid 19 after a negative antigen test then that person should be tested again with a pcr test. Based on this information half or more of covid 19 infected patients might be missed by such tests depending on the group of patients tested the who said.




















































/cdn.vox-cdn.com/uploads/chorus_asset/file/19936815/POSITIVE_COVID_19_MODEL.jpg)